ADAURA - adjuvant osimertinib reigns supreme
Recurrence rates for stage three lung cancer can be as high as 63% despite living in an age of targeted therapy and immunotherapy (1). A recent update from March 23’ on the ADAURA trial highlights promising results with significantly improved overall survival compared to placebo in those with an EGFR (epidermal growth factor receptor) mutated non-small cell lung cancer (NSCLCa) treated with adjuvant Osimertinib (Tagrisso). This is the first phase three trial to identify benefits in the adjuvant setting of targeted treatment with early stage (II and IIIA) NSCLCa after curative intent tumour resection.
The ADAURA trial, initially published in the New England Journal of Medicine (NEJM) in 2020, investigated the use of Osimertinib in phase three, a double-blinded randomised controlled trial comparing 80mg Osimertinib vs placebo for three years after patients were treated with curative surgical intent for their lung cancer(2). They had to be early-stage lung cancer (stage IB to IIIA). The primary endpoint was DFS (disease-free survival), and the secondary endpoints included overall survival, safety, and disease-free survival.
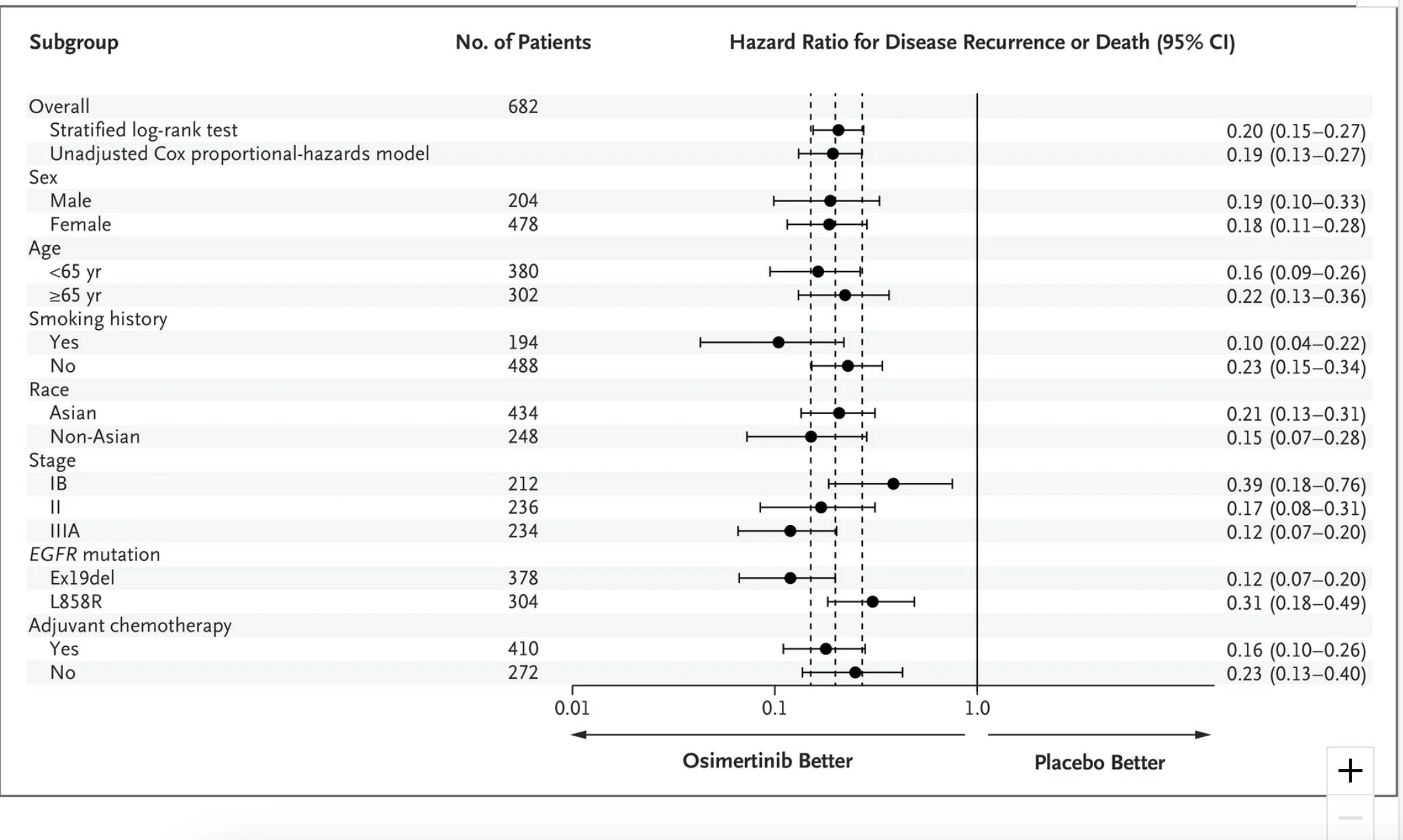
As a large trial, over 200 centres enrolled, and 682 patients were recruited across five continents. At 24 months, the primary endpoint showed that 90% of patients with stage II and IIIA resected lung cancer treated with the intervention arm (Osimertinib) and 44% in the placebo arm were disease free and alive. The hazard ratio for disease recurrence or death was 0.17 (P<0.001), a very significant and promising result. The overall population confirmed that 89% vs 52% were disease free and alive at 24 months, with an impressive hazard ratio of 0.20, favouring Osimertinib(P<0.001).
NEJM 2020
NEJM 2020
The updated results highlighted in late 2022 through the European Society for Medical Oncology Congress confirmed a median disease-free survival of 65.8 months vs 21.9 months, with a corresponding hazard ratio of 0.27, reflecting a 73% reduction in the risk of disease recurrence or death in the overall population of this trial. The placebo arm had a DFS of 38%.
Principal investigator Roy S. Herbst, MD, PhD this year released information at a press conference: "New survival data for Osimertinib reinforce the unprecedented ADAURA disease-free survival results and confirm its potential to extend patients’ lives in early stage disease”. In summary, they discussed how it was the best option for a potential cure for early-stage EGFR-mutated NSCLCa.
Historically, non-mutated stage 3 NSCLCa has been treated with chemoradiotherapy followed by maintenance durvalumab based on the results of the PACIFIC trial from 2017(3). A posthoc analysis completed last year could not conclusively identify if duravlumab was beneficial in the EGFR mutated setting. Another crucial unanswered question is whether Osimertinib has the same promise in patients treated with chemoradiotherapy rather than surgical resection as was the requirement of the ADAURA study. And finally, to add uncertainty to the realm of tomorrow, what does a clinician do if the patient recurs many years later? Is rechallenging an option?
(2) https://www.nejm.org/doi/full/10.1056/NEJMoa2027071
(3) https://www.nejm.org/doi/full/10.1056/nejmoa1709937